 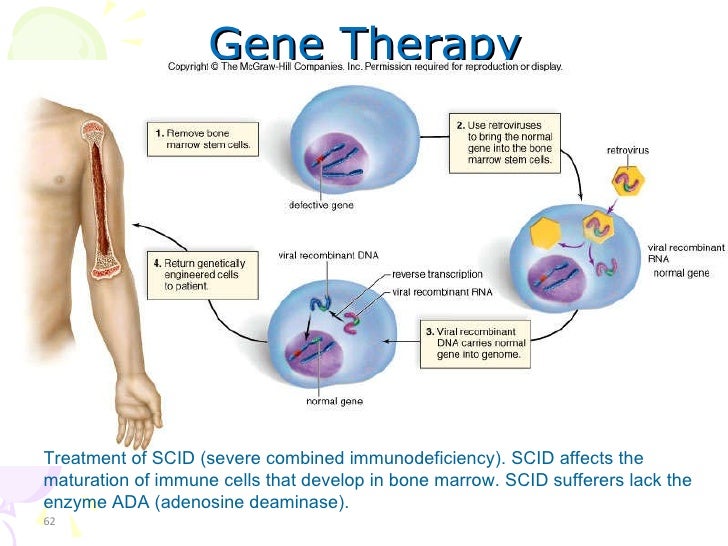 
4, 15, 16 However, to gain relevant information on the influence of vector integrations on transcriptional activity of nearby genes, studies should be carried out in relevant primary cells at the level of a clonal population containing defined insertion sites. As a result of this bias, integrations that alter the expression of flanking genes involved in cell cycle, transcriptional activity, and signal transduction might influence the biologic fate of the affected cell clone, thereby conferring a growth or survival advantage. 8-14 These studies have shown that RVs integrate nonrandomly in the host genome favoring transcription start sites (TSSs) and expressed genes, which makes productive interactions between the vector and host transcriptional machinery more probable. The genomic features driving the preferential integration of RVs have been described in several large-scale survey of mapping studies in mice, nonhuman primates, and humans enrolled in clinical trials. The trials described herein have been registered at as #NCT00598481 and #NCT00599781. These results confirm that RV-mediated gene transfer for ADA-SCID is safe, and provide crucial information for the development of future gene therapy protocols. Nonetheless, affected clones maintained a stable phenotype and normal in vitro functions. 
Quantitative transcript analysis of 120 genes within a 200-kb window around RV integration sites showed modest (2.8- to 5.2-fold) dysregulation of 5.8% genes in 18.6% of the T-cell clones compared with controls. Analysis of RV integration sites indicated a high diversity in T-cell origin, consistently with the polyclonal T-cell receptor-Vβ repertoire. T-cell clones harbored either 1 (89.8%) or 2 (10.2%) vector copies per cell and displayed partial to full correction of ADA expression, purine metabolism, and T-cell receptor-driven functions. To assess the effect of vector integration on gene expression at the single-cell level, primary T-cell clones were isolated from 2 patients. Expression profiling on ex vivo T-cell bulk population revealed no difference with respect to healthy controls. We evaluated the genomic impact of RV integration in T lymphocytes from adenosine deaminase-deficient severe combined immunodeficiency (ADA-SCID) patients 10 to 30 months after infusion of autologous, genetically corrected CD34 + cells. Gene transfer into hematopoietic stem cells by γ-retroviral vectors (RVs) is an effective treatment for inherited blood disorders, although potentially limited by the risk of insertional mutagenesis. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
